The ENFit Connector transition is coming. Read Moog Medical’s ENFit timeline update letter, or see our ENFit resources below for additional information.
For Patients & Caregivers: Your enteral feeding tube must be compatible with ENFit Connectors in order to function properly. Speak with your healthcare provider to make sure you are ready for the change.
For Homecare Providers: Do you have the proper inventory of ENFit compatible products? Have you communicated to your customers that the transition is taking place? If you need assistance, please contact us.
ENFit Enteral Connector
In an effort to prevent tubing misconnections, the International Organization for Standardization (ISO) developed the ISO 80369-3 enteral connector standard. ENFIt is a global initiative that was developed to reduce the likelihood of misconnections and accidental disconnection by creating a unique design that does not allow connectivity with connectors for any other clinical use. We have put this resource page together to ensure that clinicians, nurses, patients, and caregivers have access to information that will help ensure this transition is as smooth as possible.
What is ENFit and why was it created?
Traditional, or legacy, enteral connectors use a universal stepped adapter (left) to connect to feeding tubes. This means there is the possibility for an inadvertent connection between an enteral feeding system and a non-enteral system.
ENFIt is a global initiative that was developed to reduce the likelihood of misconnections and accidental disconnection by creating a unique design that does not allow connectivity with connectors for any other clinical use.
What are the benefits of ENFit?
The primary benefits of the ENFit change are enhanced patient safety and convenience.
| Standardized Connectors Standardized connectors across the industry increases familiarity in healthcare settings. |
|
| Reduce Misconnections The unique connector design helps ensure that enteral tubing and syringes are able to connect only to feeding tubes. |
|
| Reduce Accidental Disconnection The locking threads help prevent the connector from accidentally pulling away from the tubing. |
|
| Reduce Leaks The more secure fit of the ENFit connector reduces the potential for leaks at connector sites. |
How will you be impacted?
All enteral feeding products with a connector will be changed to include the ENFit connector. This includes syringes, gravity sets, pump sets, extension sets, and even feeding tubesthemselves. Nearly everyone who uses enteral feeding products will be impacted. Note that button style G-tubes will not be changing, just the extension sets that connect to them.
Moog has created several ENFit Enteral Feeding Connector resources designed to help readers understand how the upcoming industry-wide transition to ENFit-only products may impact them. Download the files below.
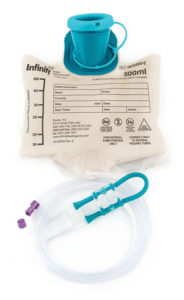
Infinity ENFit-only delivery sets
Did you know that there are Infinity enteral delivery sets currently available with only an ENFit enteral feeding connector? Click the image to learn more.
Do you have additional ENFit questions?
Contact us today and get answers from our clinical experts.
Ask our clinical experts your ENFit-related question, or simply stay up-to-date with the latest ENFit news by providing your information below.
Industry-leading clinical and customer support options right at your fingertips.
From 24 hour per day clinical support, to live stream training, self-paced e-Learning, and onsite education in your facility; our clinical and customer support teams offer the most comprehensive technical training and support in the industry. Call us at 1.800.970.2337 or click the button below for assistance. You can also find online support with our training videos, troubleshooting, FAQs, and download library.