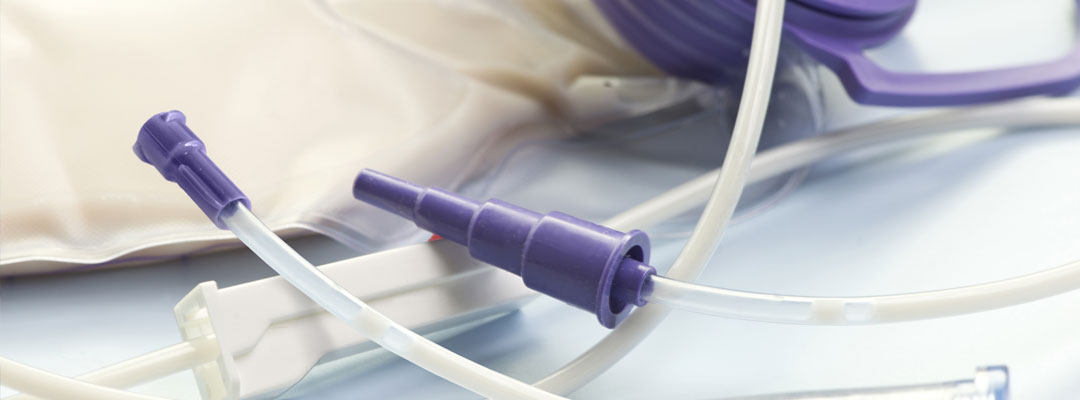
ENFit is a worldwide initiative to make all enteral feeding devices specific to tube feeding. All enteral feeding products with a connector will be changed to include the ENFit connector. This includes syringes, gravity sets, pump sets, extension sets, and even feeding tubes themselves. Nearly everyone who uses enteral feeding products will be impacted.
- Visit our ENFit information page to find answers to your questions.
This initiative, which has been spearheaded by an organization named GEDSA (Global Enteral Devices Supplier Association), was created in an effort to prevent tubing misconnections. The ENFit connector is a part of this initiative.
ENFit presents several unique benefits over traditional enteral connectors. These benefits include a standardized connector that can be used across the industry, a unique connector design that helps ensure enteral tubing and syringes are able to only connect to enteral feeding tubes, and reduced accidental disconnections and/or leaks.
There are several important dates that are coming up as the industry prepares for full transition to ENFit connectors:
- July 1st, 2021 “Legacy feeding tubes and cross application adapters will no longer be manufactured.” This means that any enteral extension set or feeding tube without an ENFit connector will no longer be manufactured after this date.
- January 1st, 2022 “Transition sets and adapters sold separately from other devices will no longer be manufactured.” This means all feeding sets (both gravity and pump set) will no longer be manufactured with the transition adapter after this date, and will only have ENFit connectors.
It is important for patients, caregivers, and clinicians to understand how they will each be impacted by the changed to ENFit connectors. Moog has created the infographic below to provide a background of why ENfit was created, what the benefits of ENFit are, how different groups are impacted by the transition to ENFit, and what is coming next as part of this transition.
Several additional resources are available to provide additional information on ENFit.

Director of Clinical Development - Enteral
Complete this form and our clinical team will contact you soon to answer your questions and provide related information and updates.
If you are experiencing issues with any of our products, please contact us using our customer support form instead.